Match 16th 2021, In accordance with the requirements of the Center for Drug Evaluation (CDE) of the National Medical Products Administration, Shenzhen Sciencare Medical Industries Co., Ltd. held a summary report on the Phase II clinical trial of naltrexone implant in Beijing by video conference.
At the meeting, Professor Hao Wei, President of the Chinese Association for Drug Abuse Prevention and Chief Clinical Researcher of Sciencare Medical, gave a keynote presentation. Former Deputy Director of the Institute of Toxicology and Medicine of the Academy of Military Medical Sciences and Chief Scientist of Sciencare Medical Professor Li Jin, formerly China Pharmaceutical University Professor Ping Qineng, Dean of the School of Pharmacy and General Counsel of Sciencare Medical, made a supplementary report and answered questions. Mr. Yin Shugui, Chairman and General Manager of Sciencare Medical, Mr. Wang Shiqiang, Deputy General Manager and Director, Mr. Zhang Tao, Executive Deputy General Manager and Director, and Mr. Qu Wei, Deputy General Manager and R&D Director, attended the meeting.

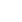
Professor Hao Wei (third from left), President of the Chinese Association for Drug Abuse Prevention and Treatment, Chief Clinical Investigator of Sciencare Medical (third from left), former Deputy Director of the Institute of Toxicology and Drugs of the Academy of Military Medical Sciences, and Chief Scientist of sciencare Medical, Professor Li Jin (second from right), former Chinese Medicine Department Professor Ping Qineng, Dean of the University School of Pharmacy and General Counsel of sciencare Medical (first from left), Mr. Yin Shugui, Chairman and General Manager of sciencare Medical (second from left), Deputy General Manager and Director, Mr. Wang Shiqiang (first from right), Executive Deputy General Manager, Director Zhang Tao (second from right), Deputy General Manager and R&D Director Mr. Qu Wei (first from left) attended the meeting.
Drugs are a worldwide public hazard that all mankind face together. It is a difficult problem to prevent and re-use after detoxification. At present, domestic experts agree that drug addiction is a kind of chronic recurrent brain disease with compulsive drug seeking and drug use behavior regardless of the consequences. The relapse rate of heroin and other opioid drug addicts after detoxification is estimated to be over 95%. Drug addicts are both offenders and victims. With the severe drug situation and the expanding of drug erosion, countries are studying and exploring to find effective anti relapse treatment.
Naltrexone implant is an ultra-long release implant developed by Sciencare Medical for the treatment of opioid use disorder and alcohol addiction. As an opioid receptor antagonist, the binding activity of naltrexone and opioid receptors is more than 30 times that of heroin and other opioid drugs. Therefore, when naltrexone reaches a certain concentration in the patient's body, it can effectively antagonize most opioids Receptors, so that drugs such as heroin cannot produce euphoria, so as to achieve the therapeutic effect of preventing relapse.
The advantage of naltrexone implants is that they take effect quickly after implantation. During this period, patients will not have the opportunity to terminate drug treatment due to "repentance", and there will be no more "persistent to drug treatment" and "abandon drug treatment" mental struggles. It is a great boon for drug addicts who get rid of the fetters of drugs.
As an innovative pharmaceutical company, Sciencare Medical has been adhering to the corporate tenet of “Goodness is like water, nourishing the world” since its establishment, and its research and development goals are the pursuit of better medicinal effects, higher safety, and better quality. The first line of drug research and development in China. For a long time, drug R&D in the world has been monopolized by pharmaceutical giants in developed countries such as Europe, America and Japan, and there is a gap between the quality of listed products in my country and the international advanced level. On October 8, 2017, the General Office of the Central Committee of the Communist Party of China and the General Office of the State Council issued the "Opinions on Deepening the Reform of the Review and Approval System and Encouraging Innovation in Drugs and Medical Devices" to encourage drug innovation and improve the competitiveness of the pharmaceutical industry to meet the clinical needs of the public. Driven by the national science and technology innovation strategy, Sciencare Medical has accumulated years of experience in research and development in the field of drug treatment and accelerated the development of drug treatment drugs.

There is no end to exploration and innovation, and drug rehabilitation is a sunrise career to save the disadvantaged. The successful convening of the second phase clinical trial summary report meeting has laid a good foundation for the follow-up research work of Sciencare Medical, and the commercialization of the drug is just around the corner. Sciencare Medical will take the improvement of domestic drug quality as the driving force, contribute to the healthy China "2030" development plan, shoulder the responsibilities of Chinese pharmaceutical companies, help patients with sincere heart, and take on more social and people's livelihood issues.















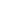


 Date:2021-03-16
Date:2021-03-16










