The naltrexone implantation independently developed by Shenzhen Sciencare Medical Industries Co., Ltd. was approved by the Center For Drug Evaluation(CDE) of the National Medical Products Administration (NMPA) and included in the "Breakthrough treatment varieties announced" List. The intended indication is to be used for the anti-relapse treatment of patients with opioid dependence after detoxification.
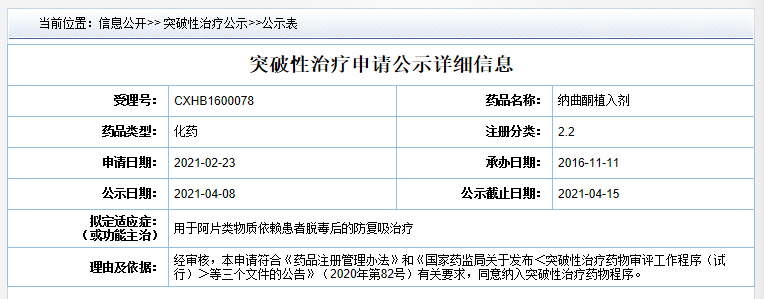
Source :Center For Drug Evaluation(CDE)website
The abuse of opioids, especially heroin, has become a worldwide public hazard. The trickiest part of opioid detoxification is relapse. According to the statistics of various countries, the relapse rate of half a year after detoxification is generally as high as 95% above.
Naltrexone is an opioid receptor antagonist, and its binding activity to opioid receptors is more than 30 times that of heroin and other opioid drugs. Therefore, naltrexone can effectively antagonize most opioid receptors when a certain concentration of naltrexone is reached in the body. Action principle of the curved ketone implants as will pay qu methadone drug molecules form evenly dispersed in the release of a particular medium, is administered by subcutaneous implant, slow balance to release drugs, for a long time to occupy the opioid receptors, antagonism opioids produced by euphoria, step by step to recover the normal distribution of synapses and neurotransmitters, finally lift the psychological dependence and resist opioids have relapsed.
In order to encourage research and development of drugs with obvious clinical advantages, CDE issued the <<Breakthrough Therapeutics Drug Evaluation Procedure (Trial)>>. For drugs included in the Breakthrough Therapeutics Drug Program, CDE will prioritize the allocation of resources to communicate, strengthen guidance and promote drug development. And, after the evaluation meets the relevant conditions, the priority to review and approve the application. The process will help accelerate drug development and product marketing for naltrexone implants.
The inclusion of Naltrexone in the Breakthrough Therapy category is a significant milestone for Sancom, and we look forward to the early introduction of new treatment options for patients in China and around the world.
About Shenzhen Sciencare Medical Industries Co., Ltd.
Shenzhen Sciencare Medical Industries Co., Ltd. is an innovation-driven biopharmaceutical companies, adhering to the "good if water Kang Ze world "of the brand concept, with" help "addiction to restart a better life in the beginner's mind, focus for addiction to develop innovative drug addiction treatment, on the basis of fully assess addiction patients needs, creative breakthrough existing mode of treatment, We are committed to improving the accessibility of drugs for addiction treatment, so that more patients can enjoy the therapeutic effects of innovative drugs. With professional team, rich pipeline, strong clinical development-driven business model and abundant capital, the company focuses on multiple product lines such as antidepressant, analgesia and oncology to further enhance the company's R&D competitiveness.
Forward-looking Statements of Sciencare Medical
Forward-looking statements made herein relate only to events or information as of the date on which such statements are made. Except as required by law, we have no obligation to update or publicly revise any forward-looking statements and unanticipated events after the date on which we make them, regardless of whether new information, future events or other circumstances arise. Please read this article carefully and understand that our actual future results or performance may differ materially from expectations. All statements in this article are made as of the date of publication and are subject to change due to future developments.

















 Date:2021-04-27
Date:2021-04-27










